Table of Contents
Marketplace Item
The CEAS is available on the Second Life marketplace along with the demonstration boards.
Introduction
The heart contains different kinds of cells that generate, conduct and respond to electrical impulses. While the pacemaker cells produce a rhythmic electrical signal in the sinoatrial node (SA) node, the cells of the conduction system transmit it to the atrial and ventricular myocardium. There, the cardiac myocytes take over in order to propagate the stimulus as a specific type of electrical pulse to its neighboring cells, the Action Potential (AP), and finally, contract. In this way, an impulse generated in the pacemaker leads to the contraction of the ventricles and the atria of the heart. The AP is a short but well described electrical event during which, each cell’s membrane potential rapidly rises and then falls, forming a typical pattern. The AP curve consists of 5 phases. The cells of the working myocardium contain fast voltage gated 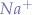 channels that permit rapid influx of sodium ions at the beginning of the AP (phase 0 or depolarization phase). This is caused by the transmission of stimuli from the surrounding cells and alters the membrane potential from approximately -90mVolts (resting membrane potential) to approximately +50mVolts. The inactivation of these channels leads to the next, short, phase 1. The “plateau” or phase 2 is the result of a
channels that permit rapid influx of sodium ions at the beginning of the AP (phase 0 or depolarization phase). This is caused by the transmission of stimuli from the surrounding cells and alters the membrane potential from approximately -90mVolts (resting membrane potential) to approximately +50mVolts. The inactivation of these channels leads to the next, short, phase 1. The “plateau” or phase 2 is the result of a 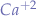 inward and
inward and  outward movements through the membrane's potassium and L-type calcium channels. This phase prolongs the AP duration on purpose: In this way the first stimulated cells of the myocardium are still in refractory state when the AP reaches the last cells of the myocardium. This prevents cyclic re-entries of APs. Phase 3 (repolarization phase) occurs when
outward movements through the membrane's potassium and L-type calcium channels. This phase prolongs the AP duration on purpose: In this way the first stimulated cells of the myocardium are still in refractory state when the AP reaches the last cells of the myocardium. This prevents cyclic re-entries of APs. Phase 3 (repolarization phase) occurs when  outward increases and
outward increases and 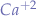 channels are deactivated. This leads the membrane back to its resting potential (phase 4). The duration of ventricular AP ranges from 200 to 400 milliseconds, while the resting heart rate is
channels are deactivated. This leads the membrane back to its resting potential (phase 4). The duration of ventricular AP ranges from 200 to 400 milliseconds, while the resting heart rate is  -
- beats per minute, influenced both by the SA node rhythm and autonomic nerves.
beats per minute, influenced both by the SA node rhythm and autonomic nerves.
Abnormal behavior of the ion channels, disturbances in the propagation among the cells or impulses originating from ectopic focuses lead to arrhythmias. For example, premature ventricular contractions are caused by APs initiated from an ectopic focus in the ventricle. As a result, the tissue depolarizes and contracts before it normally would. In ventricular fibrillation, various areas of the ventricle are excited and contract asynchronously. The long-QT syndrome is a result of delayed myocyte repolarization which may be due to mutations of ion channels or electrolyte disturbances (low blood levels of 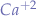 or
or  ).
).
A way of mathematically describing an excitable tissue's activity is by using cellular automata. Each cell is considered to be an automaton with a finite number of states. The transitions between the states are governed by rules resulting from basic physiology knowledge of the cardiac cell’s AP concepts. Simulation of excitable tissues based on cellular automata have been described by using various models of diffusion. One recent and promising technique aiming to reduce the complexity of reaction-diffusion systems while incorporating the simplicity of the automata theory is the Hybrid Automata (HA). Bartocci et al have developed a computer simulation environment for excitable cell networks based on HA. Such an approach is friendly both to programmer and user. We believe that cellular automata can be easily used in order to create simple but yet descriptive simulations of the electrical propagation in the heart for use in medical education.
We present Cardiac Electrical Activity Simulator(CEAS), a handy and sophisticated tool for building and studying cardiac myocyte networks in Second Life. CEAS is coded in LSL and is based on cellular automata theory, in particular on a model described by Atienza F.A. et al. which represents the electrical propagation based on probabilistic parameters.
Videos
| Normal Activity | Propagation with Dead Cells | Propagation with Re-entrance |
|---|---|---|
These three demonstration boards can be found on the SecondLife Marketplace.
Index
For the contact, copyright, license, warranty and privacy terms for the usage of this website please see the contact, license, privacy, copyright.



